LATISSE® for Eyelashes in Portland

LATISSE® (bimatoprost ophthalmic solution) 0.03% is a prescription treatment for hypo-trichosis and is used to grow eyelashes, making them longer, thicker, and darker. Eyelash hypo-trichosis is another name for having inadequate or not enough eyelashes. LATISSE® is the only FDA-approved product and has been studied for safety and efficacy and is available only by prescription. It is believed to work by increasing the growth cycle (anagen) phase of your eyelash hair cycle. The anagen is the growth phase of all hair. We believe that it increases the length of the anagen phase and the number of hairs in this growth phase.
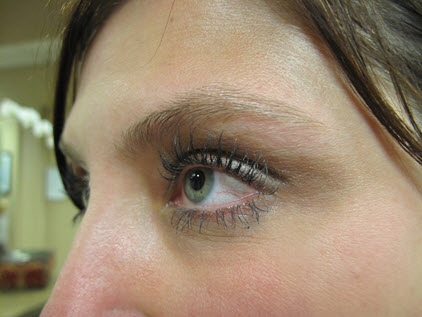 Clinical studies showed that Latisse increased lash length by 25%, (vs. 2% for vehicle); increased the fullness and thickness of the lashes by 106%(vs. 12% for vehicle) and increased lash darkness by 18% (vs. 3% for vehicle).
Clinical studies showed that Latisse increased lash length by 25%, (vs. 2% for vehicle); increased the fullness and thickness of the lashes by 106%(vs. 12% for vehicle) and increased lash darkness by 18% (vs. 3% for vehicle).
How to Apply LATISSE®
First remove any makeup and clean the upper eyelids. Remove your contact lenses, if you have them and don’t put them back in for at least 15 minutes following the application of LATISSE®. Using the single-use applicator brush provided in the package, one drop of LATISSE® is placed on the applicator brush and applied to the base of the upper eyelid lash margin.
DO NOT APPLY LATISSE® in your eye or the lower lash line as excess hair growth outside the treatment area may occur. The same process is repeated using a fresh applicator brush to avoid cross contamination. Be careful not to allow the tip of the bottle or applicator to contact surrounding structures, fingers or any other unintended surface in order to avoid contamination by common bacteria known to cause infections. After applying the LATISSE® to the lashes, blot off the excess fluid with a tissue. You will see the best results if you use LATISSE® once a day for 16 weeks. Using LATISSE® more than once a day will not grow your lashes faster or improve the effectiveness of the product.
Side effects of LATISSE® include light sensitivity and redness of the eye in less than 4% of patients who used LATISSE® in the FDA studies. There is a small possibility of developing temporary pigmentation on the upper eyelids and brown darkening of the iris of the eye, which may be permanent.

